- Continue onboarding new commercial partners, while enhancing the support to existing customers
- Launch complimentary product to Follicopeptide® Gel serum
- Continued production scale-up
- Extend product portfolio into adjacent cosmetic application ares (eyelash/eyebrow)
How it works
Follicopeptide is a modified segment of osteopontin – a natural human protein essential for healthy hair growth. It activates key processes in and around the hair follicle by stimulating stem cells, promoting new blood vessel growth, and supporting the surrounding tissue.
Androgenetic alopecia, the most common form of hair loss, occurs when hair follicles gradually miniaturise and become inactive, often due to reduced blood flow. Follicopeptide revitalises blood vessels around the follicle, enhancing growth and follicle health.
Follicopeptide
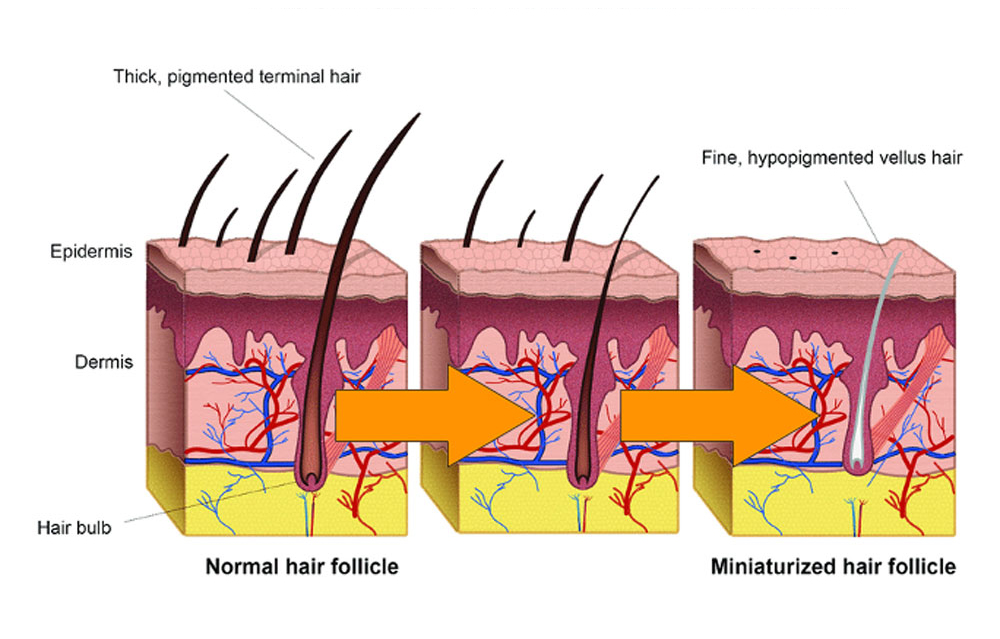
Progression of androgenic alopecia
Clinically validated
Clinical studies including more than 300 participants with androgenic alopecia demonstrated that Follicopeptide, applied once daily, resulted in remarkable growth of new hairs in individuals with a hair density of less than 255 hairs per square centimeter after just four months.